Chemistry 1 worksheet classification of matter and changes answer key – Chemistry 1 Worksheet: Classification of Matter and Changes Answer Key provides a comprehensive overview of the fundamental concepts related to the classification of matter and the changes it undergoes. This worksheet serves as a valuable resource for students seeking to enhance their understanding of this topic, offering clear explanations, engaging examples, and concise answers to commonly asked questions.
The content of the worksheet encompasses a wide range of topics, including the classification of matter into solids, liquids, and gases, the exploration of physical and chemical changes, the examination of mixtures and solutions, and the investigation of elements, compounds, and chemical reactions.
Each section provides a detailed explanation of the concepts, supported by illustrative examples, to facilitate a deeper understanding of the subject matter.
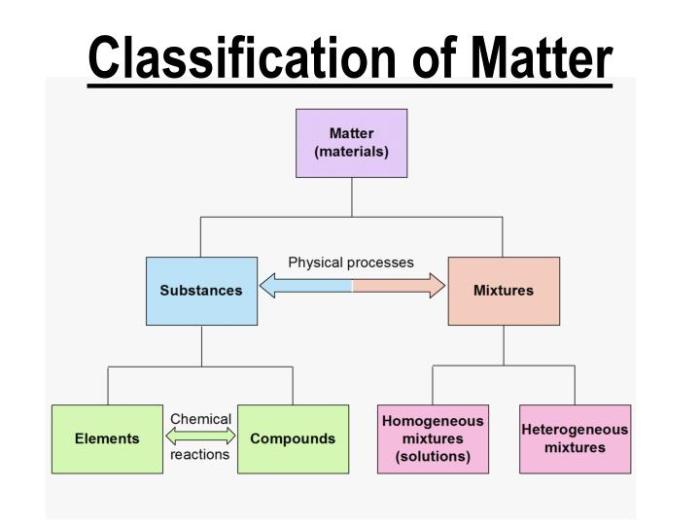
Classification of Matter
Matter exists in three states: solid, liquid, and gas. Solids have a definite shape and volume, liquids have a definite volume but no definite shape, and gases have no definite shape or volume. The properties of matter are determined by the arrangement and motion of its particles.
- Solids: In a solid, the particles are closely packed together and have little freedom of movement. This gives solids their rigidity and definite shape.
- Liquids: In a liquid, the particles are still closely packed together but have more freedom of movement than in a solid. This gives liquids their fluidity and ability to take the shape of their container.
- Gases: In a gas, the particles are far apart and have a lot of freedom of movement. This gives gases their ability to expand to fill their container.
Matter can change from one state to another by adding or removing energy. For example, adding energy to a solid can melt it into a liquid, and adding more energy can vaporize it into a gas. Removing energy from a gas can condense it into a liquid, and removing more energy can freeze it into a solid.
Physical Changes, Chemistry 1 worksheet classification of matter and changes answer key
A physical change is a change in the form or appearance of a substance without a change in its chemical composition. Examples of physical changes include melting, freezing, boiling, sublimation, and condensation.
- Melting: Melting is the change of a solid to a liquid by adding energy.
- Freezing: Freezing is the change of a liquid to a solid by removing energy.
- Boiling: Boiling is the change of a liquid to a gas by adding energy.
- Sublimation: Sublimation is the change of a solid directly to a gas without passing through the liquid state by adding energy.
- Condensation: Condensation is the change of a gas to a liquid by removing energy.
Physical changes are reversible. For example, a liquid can be frozen into a solid and then melted back into a liquid.
Chemical Changes
A chemical change is a change in the chemical composition of a substance. Examples of chemical changes include burning, rusting, and cooking.
- Burning: Burning is a chemical change that occurs when a substance reacts with oxygen to produce heat and light.
- Rusting: Rusting is a chemical change that occurs when iron reacts with oxygen and water to produce iron oxide.
- Cooking: Cooking is a chemical change that occurs when food is heated, causing the chemical composition of the food to change.
Chemical changes are not reversible. For example, once a piece of paper is burned, it cannot be turned back into paper.
Mixtures
A mixture is a combination of two or more substances that are not chemically combined. The substances in a mixture retain their individual properties. There are two types of mixtures: homogeneous and heterogeneous.
- Homogeneous mixtures: In a homogeneous mixture, the components are evenly distributed throughout the mixture. This means that the mixture has the same composition and properties throughout.
- Heterogeneous mixtures: In a heterogeneous mixture, the components are not evenly distributed throughout the mixture. This means that the mixture has different compositions and properties in different parts of the mixture.
Examples of mixtures include:
- Homogeneous mixtures: Salt water, air, milk
- Heterogeneous mixtures: Sand in water, oil and water, salad
Solutions
A solution is a homogeneous mixture of two or more substances. The substance that is present in the greatest amount is called the solvent, and the substance that is present in the lesser amount is called the solute.
- Solvent: The solvent is the substance that dissolves the solute.
- Solute: The solute is the substance that is dissolved in the solvent.
Examples of solutions include:
- Salt water: The solvent is water, and the solute is salt.
- Air: The solvent is nitrogen, and the solute is oxygen.
- Milk: The solvent is water, and the solute is fat.
Elements and Compounds
An element is a substance that cannot be broken down into simpler substances by chemical means. Elements are the building blocks of all matter. There are 118 known elements, each with its own unique properties.A compound is a substance that is composed of two or more elements that are chemically combined.
Compounds have different properties than the elements that make them up.Examples of elements and compounds:
- Elements: Hydrogen, oxygen, carbon, iron
- Compounds: Water, carbon dioxide, salt, sugar
Chemical Reactions
A chemical reaction is a process in which one or more substances are transformed into one or more different substances. Chemical reactions are represented by chemical equations. A chemical equation shows the reactants (the substances that are transformed) on the left-hand side and the products (the substances that are formed) on the right-hand side.
- Reactants: The reactants are the substances that are transformed in a chemical reaction.
- Products: The products are the substances that are formed in a chemical reaction.
Examples of chemical reactions:
- Combustion: Combustion is a chemical reaction that occurs when a substance reacts with oxygen to produce heat and light.
- Rusting: Rusting is a chemical reaction that occurs when iron reacts with oxygen and water to produce iron oxide.
- Photosynthesis: Photosynthesis is a chemical reaction that occurs when plants use sunlight to convert carbon dioxide and water into glucose and oxygen.
Stoichiometry
Stoichiometry is the study of the quantitative relationships between the reactants and products in a chemical reaction. Stoichiometry can be used to calculate the amount of reactants and products that are needed or produced in a chemical reaction.To balance a chemical equation, the coefficients in front of the reactants and products are adjusted so that the number of atoms of each element is the same on both sides of the equation.Examples
of stoichiometry calculations:
- Calculating the amount of reactants needed: If you know the amount of product that you want to produce, you can use stoichiometry to calculate the amount of reactants that you need to start with.
- Calculating the amount of products produced: If you know the amount of reactants that you have, you can use stoichiometry to calculate the amount of products that you will produce.
FAQ Compilation: Chemistry 1 Worksheet Classification Of Matter And Changes Answer Key
What is the difference between a physical change and a chemical change?
A physical change alters the form or appearance of a substance without changing its chemical composition, while a chemical change results in the formation of new substances with different chemical compositions.
What is the difference between a homogeneous and a heterogeneous mixture?
A homogeneous mixture is one in which the components are evenly distributed throughout, while a heterogeneous mixture has components that are not evenly distributed.
What is the difference between an element and a compound?
An element is a pure substance that cannot be broken down into simpler substances, while a compound is a substance composed of two or more elements chemically combined in fixed proportions.
